Properties Estimation: solubility to water
2011.6.28
Lecture note of Dr. Hiroshi Yamamoto
The program that Pirika provide.
YMB simulator (HTML5 version 2011.6.10, need pass code to use full function)
Newest version is implemented into HSPiP ver.4(Y-Predict). If the corporate visitor want to use full version, please buy HSPiP. The Abstract of HSPiP (2013.1.18)
How to buy HSPiP
Almost all the case, solubility to water data is listed g/100g water. But some source are g/1 litter or ppm and confusing. I compiled 3667 compounds' data and check the unit, then made database. It is time consuming task! And I start analyze of this result. I hadle water solubility with logS= log (g/100g water). If I check the relationship of logS to logKow (logP, octanol-water partition coefficient). I show the result below.
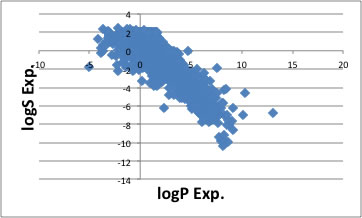
The chemicals whoes logKow is large, the nature of it is hydrophobic, so logS become small. Then I wonder why this happen. logKow is just ratio, 100/100 or 0.001/0.001 is same logP=0. But logS is amount. And logS data have another ploblem. For the compounds that dissolve more than 100g to 100g water, experiment will stop at that point. So maxium logS equal 2. With the result, additivity factor of OH or Amide become uncertain. So the estimation accuracy of this property is not so high.
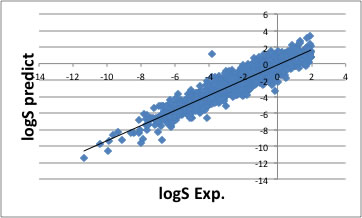
So the YMB-simulator result is not so good.
logS and logP are very important if you want to re-crystallization of chemicals or liquid-liquid extraction. I implement this scheme "bteer than nothing".
It is good idea to build this scheme for indivisual case.

